8,000+ Western blot-validated antibodies
✔ Validated on native protein from human tissues and cell lines
✔ Reproducible results with optimized protocols and support
✔ Transparent data: WB images, protocols, and HPA links for every product
✔ Enhanced Validation for over half our catalog
✔ Triple A Polyclonals™ and PrecisA Monoclonals™ data published by scientists worldwide
At Atlas Antibodies, we ensure every Triple A Polyclonal and PrecisA Monoclonal antibody is rigorously validated for Western blot applications to guarantee specific and reliable binding.
We confirm antibody binding by verifying that detected bands match the theoretical molecular weight of the target protein, while accounting for isoforms and post-translational modifications.
Our primary validation uses endogenous protein lysates from a broad panel of human tissues and cell lines. This approach closely reflects natural protein expression.
If suitable endogenous samples are unavailable, we employ recombinant full-length target proteins—commonly expressed in HEK-293 cells—to serve as positive controls.
Many antibodies are additionally validated on mouse and rat cell lysates, broadening their research applicability.
Detection is performed using peroxidase-labeled secondary antibodies combined with chemiluminescence and captured via advanced CCD camera systems, ensuring high-quality and reproducible WB imaging.
At Atlas Antibodies, we take great care to validate our antibodies and Enhanced Validation is performed as an additional layer of security in an application and context-specific manner.
The orthogonal validation method validates the antibody staining using a non-antibody-based method.
For example, the Western blot results are compared with RNA-Seq data for the same samples, using both positive and negative controls. Antibody specificity is confirmed when the antibody signal matches RNA levels in the evaluated samples.
For each antibody, two tissues or endogenous cell lines are chosen for validation: one with high RNA expression and the other with low or no RNA expression of the target protein. The cell lines are selected to express at least a five-fold difference between the RNA expression in the high and low samples.
In the enhanced validation data presented for the antibodies, the Western blot lanes in the high and low cell lines are displayed together with their corresponding RNA values.
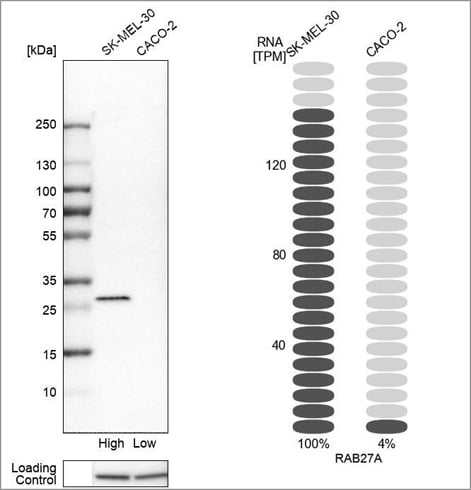
Example of orthogonal validation in WB, using the Anti-RAB27A antibody (HPA001333) in Western blot in the SK-MEL-30 and CACO-2 cell lines. On the right-hand side, bars representing the TPM values for RAB27A in the same cell lines are presented.
Genetic validation by siRNA knockdown is an enhanced method for validation where the target gene is downregulated. Antibody specificity is confirmed when the corresponding gene's knockdown levels correlate with a decrease in the antibody signal.
At Atlas Antibodies, two separate siRNA probes are employed to silence each target, and a loading control is added to ensure even loading and equal transfer over the gel. The knockdown is approved if at least 50% silencing is achieved for at least one of the two siRNA probes.
In the enhanced validation data presented for the antibody, the Western blot lanes in the control and knocked-down samples are displayed together with the loading control, and the relative remaining intensity after silencing is presented.
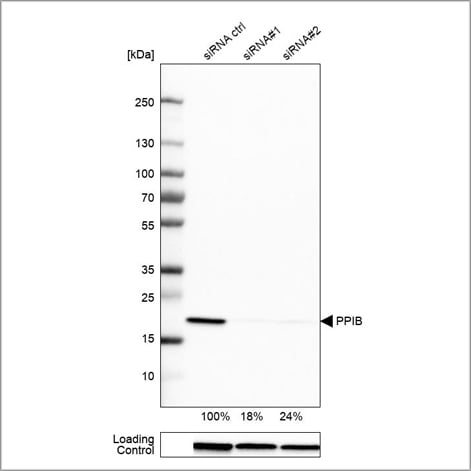
Example of genetic validation by siRNA knockdown in WB using the Anti-PPIB antibody (HPA012720). U-251 cells have been transfected with control siRNA and two target-specific siRNA probes. Downregulation of antibody signal confirms target specificity. The remaining intensity relative control lane is indicated as a percentage.
Validation by independent antibody is an enhanced method for validation where the antibody specificity is demonstrated by comparing at least two antibodies targeting the same protein with non-overlapping epitopes.
If the signals from the two antibodies correlate when compared across multiple samples, the antibodies validate each other.
In the validation data presented for the antibody, the Western blots from both antibodies are displayed together.

Example of independent antibody validation in WB using the Anti-PBLD antibody HPA038036 (A) shows a similar pattern to the independent antibody HPA038035 (B).
Recombinant expression validation is an enhanced method for validation where the antibody binding is confirmed using an over-expressed version of the target protein.
The method is applied to Western blot by comparing the antibody signal in a sample where the target protein has been recombinantly over-expressed with the signal from a control sample. Antibody specificity is confirmed when the antibody shows a strong band in the cell line with recombinant expression and no or faint band in the control line.
In the validation data presented for the antibody, the Western blot includes the over-expressed sample and the control sample in the same blot.
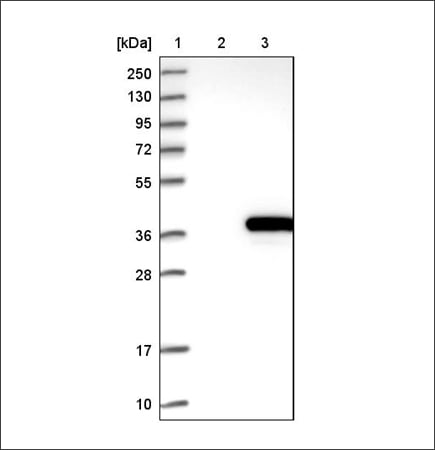
Example of recombinant expression validation in WB using the Anti-ACY3 antibody (HPA039219). Lane 1: marker, lane 2: negative control (vector only transfected HEK293T lysate), lane 3: ACY3 over-expression lysate (Co-expressed with a C-terminal myc-DDK tag (~3.1 kDa) in mammalian HEK293T cells, LY408962).
Loading controls are helpful to ensure that an even amount of protein is loaded on the gel.
When studying the expression of a target protein in different samples, it is necessary to normalize protein levels using a loading control antibody to get reliable data. By using a loading control, you can distinguish an unevenly loaded sample from an actual difference in the protein expression between the samples.
Suitable targets for loading control are housekeeping proteins expressed at high levels in a wide range of cell lines and tissues.
Loading controls can also be used to confirm that the transfer of protein from the gel is equal over the whole membrane. This is particularly useful when comparing protein expressions over several samples.
When choosing a loading control, be sure to pick one that targets a protein of a different molecular weight than that of your protein of interest. By doing so, you can easily differentiate between the two bands during the blotting.
For optimal results, also check that the protein or RNA expression level of the loading control target is fairly high in the cell lines or tissue lysates you will use.
Fluorescence-based multiplex Western blot is a time-saving alternative to chemiluminescent detection. For multiplexing, choose a loading control antibody with a different isotype or host species than the antibody targeting the protein of interest.
Using suitable secondary antibodies conjugated with fluorescent dyes, the western blot protocol can be reduced to only one primary and one secondary incubation step. The two bands can be visualized in the same image.
Discover our antibody panel of Loading Controls, consisting of PrecisA Monoclonals targeting proteins that are constitutively expressed in a large set of cell lines and tissues. The Atlas Antibodies Loading Control panel contains antibodies of different isotypes allowing multiplexing.
Browse loading control antibodies
Use these recommended protocols for optimal results in Western blot using our antibodies. The protocols are optimized for Triple A Polyclonals and PrecisA Monoclonals.
Western blot Standard Protocol
Western blot Protocol - BSA Blocking
To further confirm antibody specificity, we apply four enhanced validation methods: