Visualizing ATRX Loss: How a New 3D Glioma Model Unlocks Insights into Malignant Transformation
A Breakthrough View into Glioma Progression
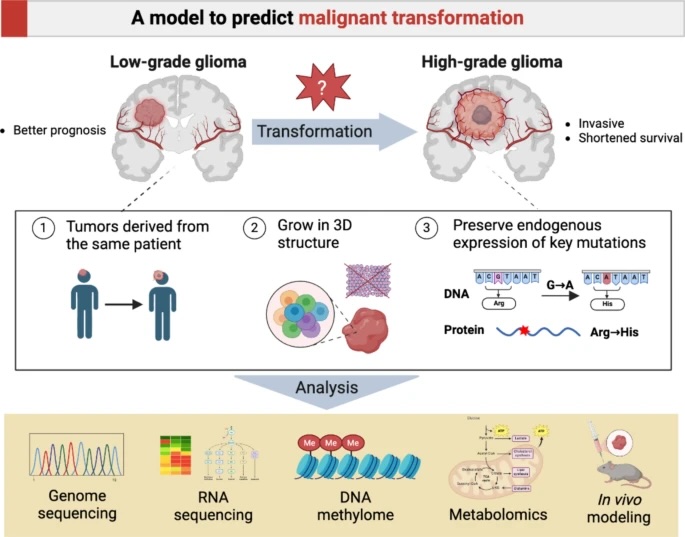
Gliomas, a type of brain tumor, are notoriously difficult to study, especially the process by which they transform from low-grade to highly aggressive cancers. A pioneering study recently published in Acta Neuropathologica Communications has developed a vital new tool to understand this malignant transformation, providing hope for future therapies.
The study, led by Kim O. et al., focuses on IDH-mutant gliomas, a common subtype. Researchers successfully created a patient-derived 3D cell model that mimics the tumor's progression from a low-grade (WHO grade 2) to a high-grade (WHO grade 4) state, a significant leap forward in cancer research.
The Power of a Patient-Derived Model
For the first time, researchers have established matched 3D cell models from a single patient's tumor at two different stages:
403L: Representing the initial low-grade glioma (LGG).
403H: Representing the subsequent high-grade glioma (HGG).
This unique "before and after" view allows for a precise comparison of the genetic, metabolic, and signaling changes that drive a tumor's aggressive turn.
To validate their model and study protein expression, the researchers used the anti-ATRX antibody (HPA001906) from Atlas Antibodies for immunohistochemistry (IHC). The loss of the ATRX protein is a key marker in glioma progression, and our antibody was crucial in confirming this feature in the patient's tissue and the new cell models.
Key Findings: What Drives Malignant Transformation?
The study revealed several critical changes that occur as the glioma becomes more aggressive.
Genetic and Proliferative Markers
Both the low-grade and high-grade models accurately expressed the IDH1 R132H mutant protein, confirming their clinical relevance.
IHC staining with the anti-ATRX antibody confirmed the loss of ATRX expression in the nuclei of both tumors, a hallmark of this cancer type.
The proliferative marker Ki-67 showed significantly stronger expression in the high-grade (403H) tumor, indicating rapid cell division and aggressiveness.
Signaling and Metabolic Shifts
Pathway analysis showed a shift from Notch signaling in the low-grade tumor to pathways promoting epithelial-mesenchymal transition (EMT) in the high-grade tumor—a key process in cancer invasion.
The tumor's metabolism changed dramatically, with the high-grade model showing elevated levels of citrate, glutamine, and the oncometabolite 2-HG, which fuel tumor growth.
Why This Research Matters for the Future
This study is more than just an academic exercise; it has powerful real-world implications.
A Vital New Research Tool: This 3D model is the first of its kind for IDH-mutant gliomas and provides an invaluable platform to test new drugs and therapeutic strategies.
Identifying Therapeutic Targets: By understanding the specific metabolic and signaling changes, scientists can now explore targeted interventions to halt or prevent the tumor's malignant transformation at an earlier stage.
Validating High-Quality Reagents: The success of this study underscores the importance of using highly validated antibodies, like our anti-ATRX antibody, to produce reliable and reproducible data in critical cancer research.
This breakthrough offers a clearer roadmap for tackling IDH-mutant gliomas and brings researchers one step closer to developing more effective treatments.
Read the full publication
Acta Neuropathologica Communications: Kim O, et al. (2024). A patient-derived cell model for malignant transformation in IDH-mutant glioma. Acta Neuropathol Commun. 12(1):148.
