At Atlas Antibodies, we know that a high-quality antibody is only part of the equation for a successful IHC experiment. As an original antibody producer with roots in the Human Protein Atlas project, we've spent decades perfecting not just our antibodies, but the protocols that make them work. Antigen retrieval is one of the most critical steps, and getting it right is the key to unlocking clear, reproducible data. In this guide, we'll share our expertise on why it matters and how to perform it correctly.
Antigen retrieval is a critical pre-analytical step in immunohistochemistry (IHC) that restores epitope accessibility following formalin fixation. Without proper unmasking, even high-quality antibodies (monoclonal or polyclonal) may fail to bind their targets, leading to common problems like weak IHC staining, high background IHC, or even false negatives.
Why Antigen Retrieval Matters
The Challenge of Formalin Fixation and Epitope Masking
Formalin fixation creates a significant challenge in immunohistochemistry by masking tissue antigens. As a cross-linking fixative that forms methylene bridges between proteins, formalin has remained the standard choice for histopathological tissue processing since 1893. The primary artifact of this fixation process is antigen masking, where cross-linking between amino acid residues alters protein structure and eliminates the ability of primary antibodies to recognize their target peptide epitopes. The discovery in 1991 that these cross-linkages could be reversed through high-temperature heating or strong alkaline treatment led to the development of modern antigen retrieval techniques.
What Is Antigen Retrieval? (The Solution to Masking)
Antigen retrieval is the process of restoring epitope accessibility by disrupting formalin-induced crosslinks through heat or enzymatic digestion. When formaldehyde crosslinks alter the three-dimensional conformation of epitopes, even high-affinity antibodies cannot bind to their targets effectively. By deliberately breaking these crosslinks, antigen retrieval exposes the binding sites and enables accurate antibody recognition. Selecting the appropriate retrieval method is crucial for achieving consistent and reliable immunohistochemical results.
How to Get Antigen Retrieval Right
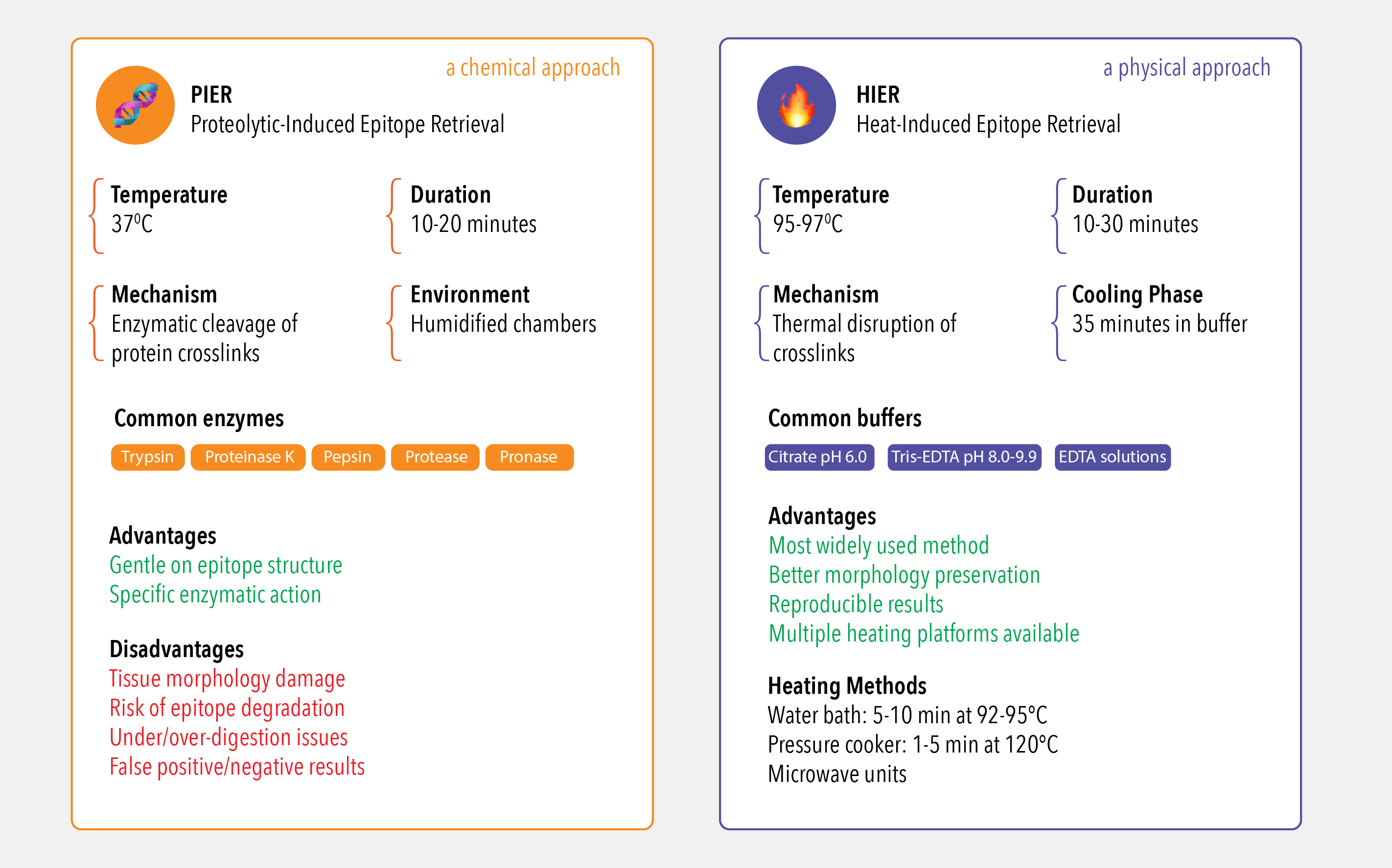
Method 1: Proteolytic-Induced Epitope Retrieval (PIER) - the chemical approach
PIER employs proteolytic enzymes to cleave protein crosslinks and restore antigenic accessibility. This method typically operates at 37°C with incubation periods of 10-20 minutes in humidified chambers. Commonly utilized enzymes include trypsin (optimally at pH 7.8), proteinase K, pepsin, protease, and pronase, each requiring specific buffer conditions for maximum efficacy.
Optimal retrieval conditions depend on tissue type, fixation method, and target antibody, necessitating systematic optimization through matrix studies combining various time, temperature, and pH parameters. However, PIER presents significant limitations including morphological tissue damage, potential epitope degradation leading to false-negative results, and the critical balance between under-digestion (insufficient antigen exposure resulting in weak immunostaining) and over-digestion (causing false-positive staining, elevated background, and structural tissue damage). Consequently, PIER is used less frequently than HIER due to these morphological and antigenic alterations.
Method 2: Heat-Induced Epitope Retrieval (HIER) - the physical approach
HIER represents the most widely used pretreatment method in IHC for formalin-fixed paraffin-embedded tissues. This technique utilizes temperatures of 95-97°C to disrupt protein crosslinks through thermal unfolding, with typical protocols involving 10-30 minute heating periods followed by 35-minute cooling phases in buffer. Temperature and timing optimization varies by heating method: 5-10 minutes at 92-95°C in water baths versus 1-5 minutes at 120°C in pressure cookers.
Buffer selection is antigen-dependent and pH-critical, with low pH citrate buffer (pH 6.0) and high pH Tris-EDTA solutions (pH 8.0-9.9) representing the most frequently employed formulations. The mechanism involves both thermal disruption of crosslinks and chelation of calcium ions from protein cross-linking. Modern HIER applications utilize diverse heating platforms including domestic and purpose-built microwave units, pressure cookers, and water bath systems, each requiring specific optimization protocols for reproducible results. At Atlas Antibodies, the vast majority of our 12,000+ IHC-validated primary antibodies are optimized using a standardized HIER protocol, ensuring you have a reliable starting point for your experiments.
Is antigen retrieval always necessary?
No, it is not. Antigen retrieval is not universally required for all immunohistochemical procedures. The necessity depends on the fixation method used, as mild fixation conditions may not mask epitopes sufficiently to require retrieval. The decision to employ antigen retrieval should be based on several key factors: the specific target antigen, the primary antibody characteristics, the tissue fixation method, and the desired staining intensity and consistency. This is why reviewing the validation data provided by a primary manufacturer like Atlas Antibodies is so crucial.
Tissue-Specific Considerations
Frozen tissues fixed with alcohol do not require antigen retrieval since alcohols do not mask epitopes. Similarly, fresh frozen sections typically require only brief fixation with ice-cold acetone at -20°C for 10-20 minutes, which preserves antigenic structures without creating extensive crosslinks. Antigen retrieval is primarily needed for fixed tissues to break cross-linkages formed through fixation, especially formalin.
Certain antigens demonstrate inherent resilience to fixation-induced masking due to their abundance, structural robustness, or resistance to crosslinking. These proteins may maintain adequate immunoreactivity without retrieval procedures, though antigen retrieval can still enhance staining consistency and intensity.
Optimization Strategy and Best Practices
A systematic approach is key to success. We recommend starting with the protocol on the Atlas Antibodies product datasheet, as it has been pre-optimized for that specific antibody. If you need to develop a protocol from scratch, your strategy should include the following:
A Systematic Approach to Optimization:
When a pre-optimized protocol isn't available, here is a simple, three-step strategy we recommend to find the optimal conditions for your antibody:
- Start with HIER at both low pH (citrate buffer, pH 6.0) and high pH (Tris-EDTA, pH 8.0-9.9)
- Evaluate PIER using different enzymatic approaches (trypsin, proteinase K, pepsin)
- Conduct preliminary matrix studies using various combinations of time, temperature, and pH to optimize retrieval conditions.
Essential Quality Controls for IHC:
Always consider the possibility of artifacts. We recommend including the following controls in every experiment to ensure your staining is specific and reliable:
Negative Controls: Sections processed without the primary antibody to check for non-specific binding from the secondary antibody.
Positive Controls: Tissues with known antigen expression to confirm that your protocol and reagents are working correctly.
Specificity Controls: Use methods like knockout/knockdown validation or blocking peptides to ensure your antibody is binding to the correct target.
The Ultimate Control: Matched Antibody & Antigen Pairs. To achieve the highest possible confidence, nothing is more powerful than using the exact antigen as a control. As an original antibody producer, Atlas Antibodies has a unique advantage: for many of our primary antibodies, we also offer the corresponding PrEST Antigen™ that was used to generate and validate it. Using this matched pair allows you to confirm your entire workflow—including your antigen retrieval step—is optimized correctly, providing a level of certainty that is simply unmatched.
Invest in Reliable Equipment:
Invest in reliable, temperature-controlled heating equipment for HIER procedures to ensure reproducible results and minimize protocol variability between experiments.
This systematic approach ensures optimal immunostaining results while minimizing artifacts and maintaining scientific rigor in antigen retrieval selection and implementation.
Troubleshooting Common Antigen Retrieval Issues
Even with a good protocol, you can run into issues. If you're seeing weak IHC staining or IHC no signal, it could be due to under-retrieval. Conversely, high background in IHC can be a sign of over-retrieval or tissue damage. Systematically testing your buffer pH and timing is the best way to resolve these IHC artifacts.
Frequently Asked Questions
Q: What is the best antigen retrieval buffer to use?
A: The optimal antigen retrieval buffer depends on the antibody. We recommend starting with both a low pH (Citrate Buffer, pH 6.0) and a high pH (Tris-EDTA Buffer, pH 9.0) to see which yields the best results.
Q: How can I fix weak IHC staining?
A: Weak IHC staining is often caused by insufficient epitope unmasking. Try increasing the heating time or switching to a higher pH retrieval solution, and ensure you are using a validated primary antibody.
Your Partner in IHC Success
Effective antigen retrieval is a foundational step in generating reproducible IHC data. Getting it right ensures that the highly validated primary antibody you've chosen can perform at its best.
As a primary antibody manufacturer, our commitment to your success goes beyond the vial. We provide not only rigorously tested, application-specific protocols, but also the tools for ultimate experimental confidence, like our matched PrEST Antigens™. By starting with a complete and well-validated system, you can turn potential artifacts into a publishable signal.
Need help with protocol optimization or buffer selection? Contact our technical support or browse validated antibodies at Atlas Antibodies.
